GeneTex
Led by the distinguished Dr. Eva Lee, GeneTex has established state-of-the-art facilities in both the United States and Taiwan to provide top-quality antibodies and reagents generated through refined product development programs, international collaborations with leading researchers, and scrupulous scientific rigor.
GeneTex creates reagents for biomedical research, assay development, and in vitro diagnostics. Its products are made to standards outlined in quality management systems ISO 9001:2015, ISO 13485:2016, and ISO 14001:2015 are supported by comprehensive customer service.

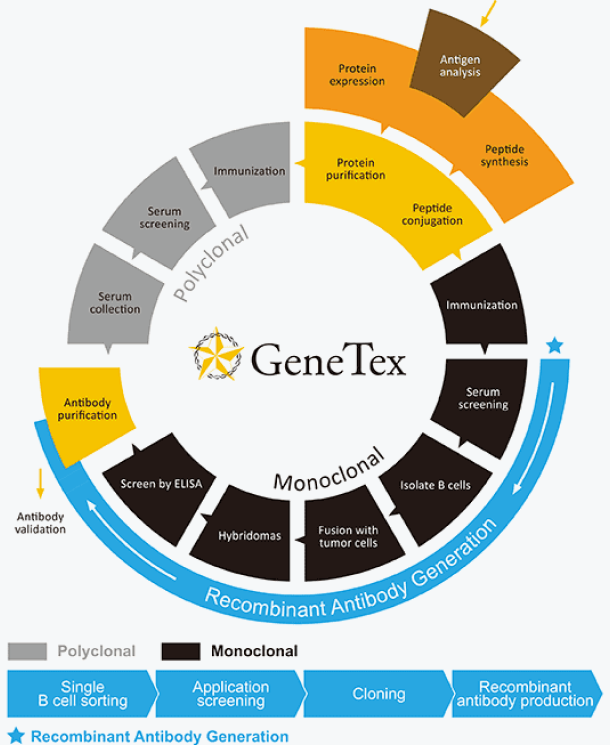
Precision and reliability in every antibody
While commercial polyclonal and monoclonal antibodies have driven progress in science and medicine since their introduction, the production of reliable antibodies remains a crucial objective in the pursuit of data accuracy and reproducibility. As a pioneer in the production of high-quality antibodies, GeneTex has successfully integrated recombinant monoclonal antibody technology into its product development platform. This technology offers numerous well-documented advantages, with consistency of performance being of paramount importance. GeneTex's capability in this area strengthens the company's position within the research community and the biotechnology industry and spearheads its efforts to deliver the most reliable antibody products available.
“Our commitment to provide our customers with reliable reagents and to drive life science research reflects the philosophy and approach we employ when we create our products.”
Using careful manufacturing processes and rigorous testing, GeneTex has produced and validated a comprehensive collection of antibodies and reagents that researchers use daily to expand their biomedical knowledge.
Ensuring antibody quality
For over 25 years, GeneTex has produced high-quality antibodies and reagents, distinguishing itself from other antibody suppliers by taking the necessary steps to carefully validate its antibodies. Poorly characterized antibodies remain an industry-wide challenge that can significantly impact progress in the life sciences and biomedicine.
GeneTex's primary concern is the reliability of its products. Rigorous validation protocols have been developed to test its antibodies for various applications, including Western blot (WB), immunoprecipitation (IP), immunohistochemistry (IHC), intracellular computed tomography/infrastructure testing (ICC/IF), FACS, and ELISA, using cell lines with known target protein expression levels and a wide variety of tissue samples. Batch consistency is ensured through a combination of pre-qualified reference batches, clone verification, and thorough quality control.
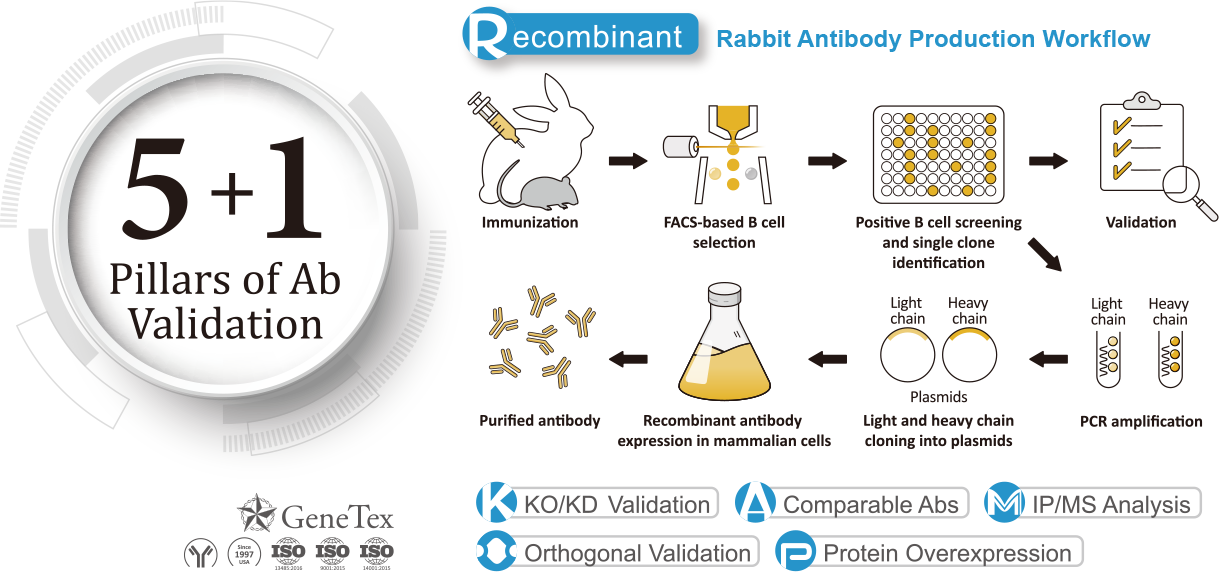
The 5+1 pillars of the antibody validation method:
To optimize the performance, quality and specificity of its antibodies, GeneTex employs various analytical validation strategies in its 5+1 pillar method.
Key GeneTex Research Areas
Infectious Disease Research Antibodies
GeneTex offers a wide range of antibodies for infectious disease research, including reagents for SARS-CoV-2, influenza, dengue virus, RSV, HIV, hepatitis viruses, adenovirus, Ebola virus, Zika virus, Mpox virus and many more. These products are suitable for researchers studying viral detection, host-pathogen interactions and assay development.
Cancer & Tumour Marker Antibodies
GeneTex provides antibodies for important cancer markers across pancreatic, breast, ovarian, liver, colorectal, melanoma, lung cancer and leukaemia research. The range includes targets such as CA 19-9, HER2/ERBB2, ER, PR, CA 125, HE4, AFP, Glypican-3, CEA and S100 beta.
Cancer Immune Checkpoint Antibodies
For immuno-oncology research, GeneTex supplies antibodies for both inhibitory and stimulatory immune checkpoint targets. Key targets include PD-1, PD-L1, CTLA4, TIGIT, TIM-3, LAG3, CD28, CD40, CD73, IDO1 and CD47. These reagents support research into tumour immune escape, checkpoint signalling and cancer immunotherapy pathways.
Neuroscience Research Antibodies
GeneTex has a strong neuroscience antibody range covering neural progenitor cells, mature neurons, astrocytes, microglia, oligodendrocytes, synapses and neurotransmitter systems. Popular targets include GFAP, NeuN, Iba1, OLIG2, MAP2, beta Tubulin 3/Tuj1, Synaptophysin, PSD95, GAD65/67 and Tau.
Neurodegenerative Disease Research
GeneTex antibodies support research into Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, ALS and prion disease. Key products target proteins such as beta Amyloid, Tau, alpha Synuclein, C9orf72, Huntingtin, SOD1, TDP43 and P62/SQSTM1.
