Neuronal Tracers
The nervous system is made up of a network of specialised synaptic connections that appear to be infinitely complex and regulate the movement of information through it. The goal of connectomics is to map the structure that underlies brain function at resolutions ranging from the ultrastructural, which looks at the arrangement of individual synapses that impinge on a neuron, to the macroscopic, which looks at the overall connectivity between large brain areas.
The synaptic connectivity patterns that are thought to underlie the functional characteristics of neural circuits are identified by neural tracers. Neuronal tracers can demonstrate the anatomical connections of neurons. They can be transported retrogradely, from the axon to the soma, or anterogradely from the soma to the axon, depending on whether the endpoint or the origin of a neuron needs to be found to outline the transport pathways in neurons. Retrograde tracing enables the "mapping" of connections between neurons in a specific structure (for example, the eye) and target neurons in the brain. Anterograde tracing, on the other hand, traces neural connections from their source to their point of termination (i.e., from cell body to synapse).
More recently, viral tracers which are direction specific, have become popular as they can be manufactured to target specific cell types as well as express small molecule tracers in the cells of interest. Viruses have the advantage of self-replication over molecular tracers. Since viruses have the ability to jump across synapses they can spread very quickly and degrade neural tissue. Viral tracers are useful tools for neuroanatomical mapping and genetic payload delivery. Unlike non-viral tracers, which have many limitations, genetically modified viruses can target specific cell types. Utilising viral vectors, and tracing multiple synapses has been made more efficient, however, they may require more stringent laboratory safety measures. (Saleeba, 2019)
This article covers reagents available from 2BScientific Limited, but for a complete review, please see the references.
Anterograde tracers
Phaseolus vulgaris Leucoagglutinin
Phaseolus vulgaris Leucoagglutinin is a specific marker for tracing efferent neuronal projections. Following iontophoretic injection, PHA-L is transported at a rate of 4-6mm/day, and a survival rate of over 18 days has been observed. PHA-L is fixable and can be detected using an Anti-PHA (E+L) antibody to the lectin. PHA-L has a molecular weight of approximately 126kDa.
Dextran Amines
Dextran amines are usually around 10kDa and can be used labelled, popularly with biotin or a fluorophore. They are introduced by iontophoretic or pressure injection methods. As they can come labelled both with the fluorophore and biotin, fluorescence can be used to monitor the injection site, and further immunohistochemical labelling via the biotin can later occur. Dextran amine labelled tissues are fixable.
Herpes Simplex Virus Type 1
HSV1 infects a broad range of cells and shows fast trans-neuronal spread in the central nervous system and can lead to meningitis or encephalitis. Multiple replication-defective or competent strains are available at 2B Scientific for research purposes.
Anterograde and Retrograde Tracers
NEUROBIOTIN™ / Biocytin
NEUROBIOTIN is an amino derivative of biotin that can be used as an intracellular label for cells, particularly neurons and some have fluorophores conjugated enabling direct visualisation as well as an alternative label via the biotin. They can be used to indicate gap junction coupling. NEUROBIOTIN tracer offers better solubility, more efficient iontophoresis, remains in the cell longer, and is non-toxic. It can also be fixed with either formalin or glutaraldehyde. Although used in similar circumstances, NEUROBIOTIN offers some advantages over biocytin.
Wheat Germ Agglutinin
Wheat germ agglutinin (WGA) contains a group of closely related isolectins, with an isoelectric point of about pH 9. As with Cholera B Toxin, conjugating HRP to WGA offers a 60-fold improvement of sensitivity compared to HRP alone. This may be partly due to the amplification of the signal as part of the detection method. As this is a bidirectional tracer, it can jump trans-ganglionically in peripheral circuits, or trans-synaptically in central circuits. It is transported relatively quickly, with reports that in adult mice the long spinocerebellar axons and their terminals in the cerebellum can be traced in their entirety in less than 6 hours from an injection.
Horseradish Peroxidase (Predominantly retrograde)
One of the initial neuronal tracers known, HRP is transported in both directions, although predominantly in the retrograde direction. They are detected simply by incubation with a suitable HRP substrate such as DAB. It must be taken care of during the treatment of the tissue not to destroy the HRP during processing though.
Lipophilic Tracers
Carbocyanine dyes are extremely lipophilic resulting in uptake and diffusion exclusively in lipid compartments such as the myelin sheath. Active transport is not involved for these dyes, so transport is bidirectional, and is suitable for use in fixed tissues as well as in living organisms. These dyes can be used in a wide variety of applications for the long-term tracing of cells, including cell transplantation, migration, adhesion, and fusion studies.
The most popular lipophilic tracers, DiO (DiOC18(3)), DiI (DiIC18(3)), DiD (DiIC18(5)) and DiR (DiIC18(7)) are weakly fluorescent in the aqueous phase, but highly fluorescent and photostable when incorporated into membranes or bound to lipophilic biomolecules.
Retrograde
Cholera toxin B Subunit
Cholera toxin B subunit (CTB) has been reported to be less rapidly eliminated from retrogradely labelled neurons than native HRP, and like WGA conjugates, offers increased sensitivity – most likely due to amplification provided by the extra layers in the detection method. When unconjugated, CTB is exclusively transported retrogradely. Fluorophore conjugates can be used to monitor transport progress. Although most work has been carried out in mammals, work in pigeons suggests that this tracer is also suitable for non-mammals.
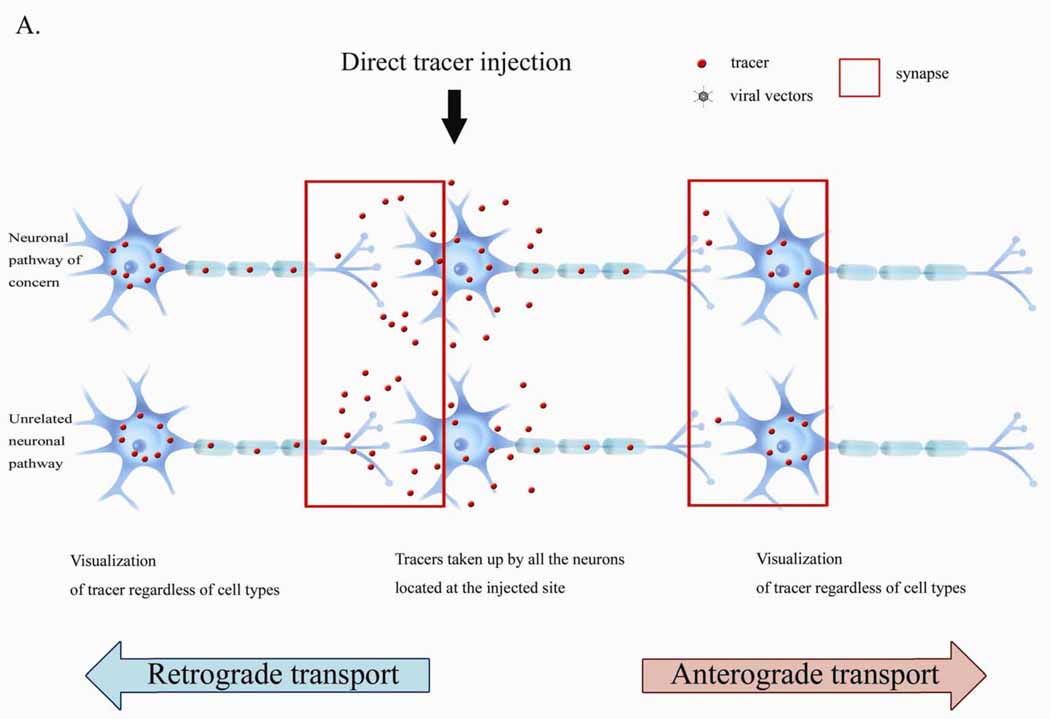
Figure 1: Conventional Tracer Delivery: Tracers can be taken up by all neurons near the injection sites, resulting in nonspecific labelling of unrelated pathways. (Huh, 2010)
Toxin Antibodies
Infects many host species and has transneuronal spread in the nervous system and CNS. It has been extensively used to map synaptic circuits in the CNS and PNS. The virus replicates only within populations of synaptically connected neurons and does not spread through extracellular space or cell-to-cell fusion.
Infecting almost all mammals, the rabies virus is a neurotrophic virus that can infect a broad range of hosts. The genetically modified rabies virus is well known as a monosynaptic tracing system. It enters the nervous system either through the neuromuscular junction of a motor neuron or the nerve spindles of a sensory nerve.
Vesicular Stomatitis Virus (VSV)
It can replicate rapidly in cells, relatively independently of the cell cycle. Modifications are available to enable tracing over one synapse or multiple synapses. It causes infection of the central nervous system when administered intranasally. When infected macrophage precursors or memory T-cells cross the BBB and differentiate into perivascular microglia.
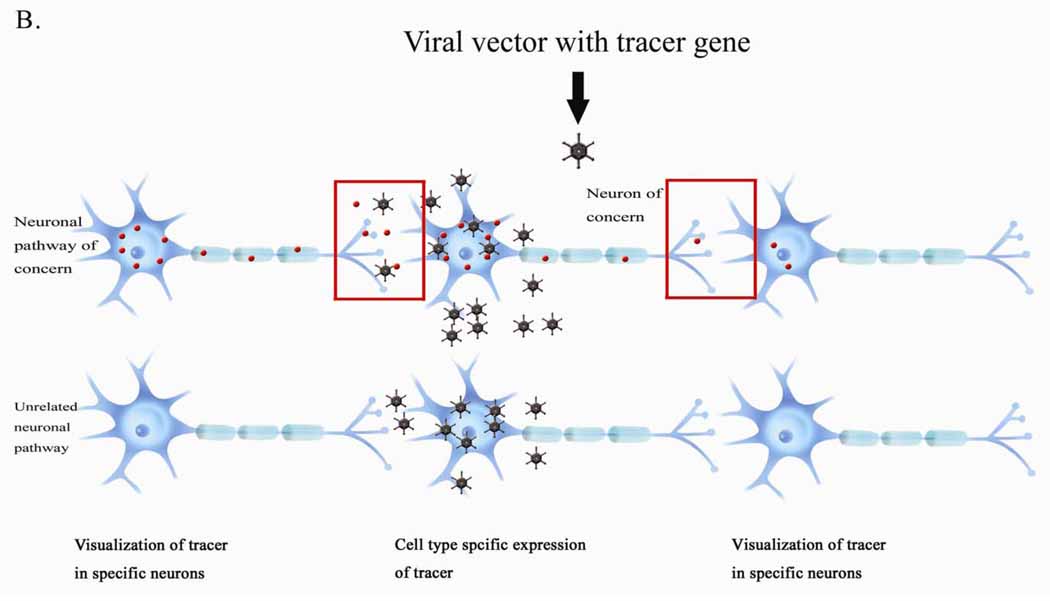
Figure 2: Viral tracer delivery: Tracers can be expressed only in particular neurons, allowing for the visualisation of trans-synaptic neuronal pathways. (Huh, 2010)
